AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Molar mass on a periodic table3/31/2024  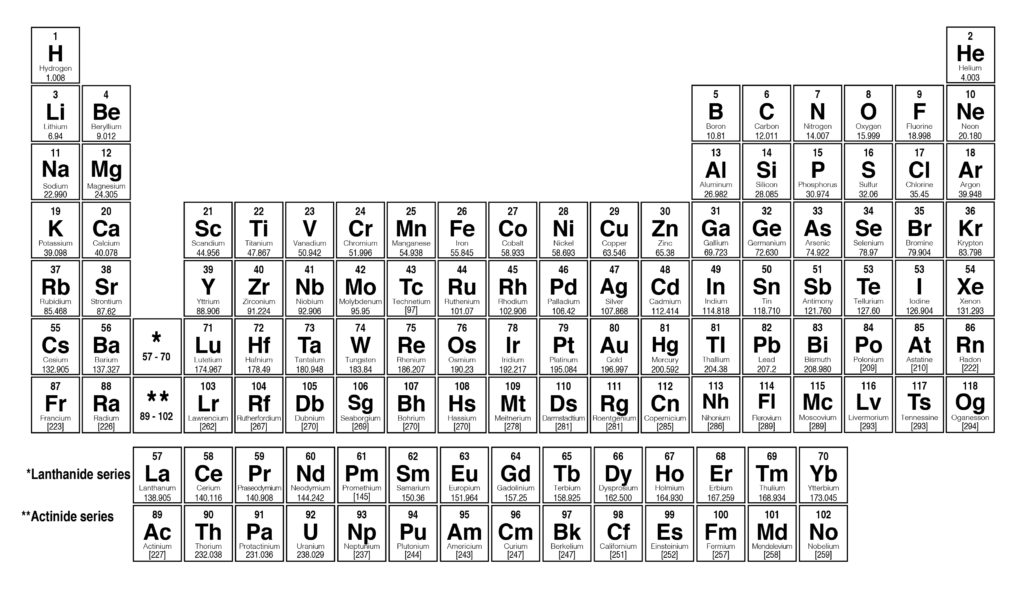
: From the formula (H2SO4), we can say the following: When you have used the periodic table to calculate the Ar values, move on to the third step. : Now, let’s use the periodic table to find the Ar values of these elements. From the formula H2SO4, we can say that this compound has three elements: : First of all, we will identify all the elements in this compound. Let me explain all this to you with the help of some examples: Multiply the number of atoms with the Ar value.Use the periodic table to find the A r value(s) of the element.To find the molar mass, we are going to do four simple steps: How to find the molar mass on the periodic table? With this, it is time to move on and talk about the calculation of molar mass. As you may know, these elements are arranged by (increasing) atomic number (number of protons in the nucleus of the atom). The periodic table of chemical elements is a table that displays the chemical elements. Similarly, the mass of 1 mole of H2O molecule is 18g.īut before telling you how to calculate the molar mass using the periodic table, let me tell you what a periodic table is. However, the numerical values of both of them are the same.įor example, the molecular mass of 1 H2O molecule is 18 a.m.u. The ions are then separated in a mass analyzer according to their mass-to-charge ratio.Molecular mass (also known as molecular weight) is also the mass of one molecule of a substance. It requires that all molecules and atoms to be measured be ionized. Mass spectrometry is a technique that measures the mass-to-charge ratio (m/q) of ions. Masses of atoms and molecules are measured by mass spectrometry. Neither u nor Da are SI units, but both are recognized by the SI. For this reason, the dalton (Da) is increasingly recommended as the accurate mass unit. Hence, the amu is no longer in use those who still use it do so with the definition of the u in mind. Therefore, both communities agreed to the compromise of using m( 12C)/12 as the new unit, naming it the "unified atomic mass unit" (u). Because the isotopic distribution in nature can change, this definition is a moving target. The amu was defined differently by physicists and by chemists:Ĭhemists used oxygen in the naturally occurring isotopic distribution as the reference.  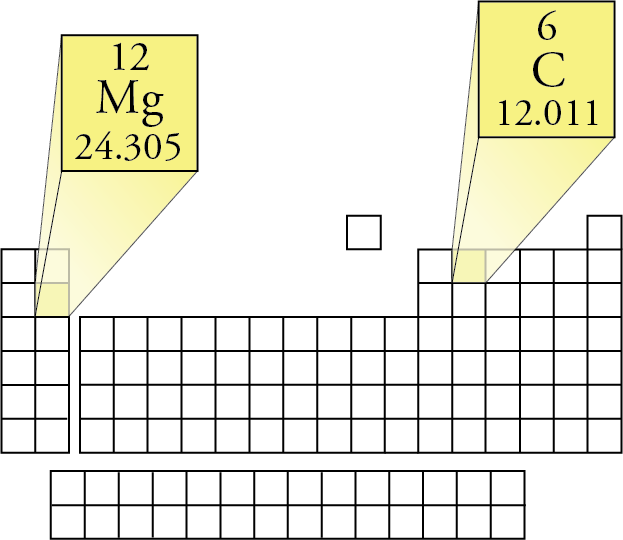
Some chemists use the atomic mass unit (amu). Berzelius demonstrated that this is not always the case by showing that chlorine (Cl) has a mass of 35.45, which is not a whole number multiple of hydrogen's mass. Known was Prout's Law, Prout suggested that the known elements had atomic weights that were whole number multiples of the atomic mass of hydrogen. Early atomic mass theory was proposed by the English chemist William Prout in a series of published papers in 18. The first scientists to measure atomic mass were John Dalton (between 18) and Jons Jacoband Berzelius (between 18). So 1 u is 1/12 of the mass of a carbon-12 isotope: Both units are derived from the carbon-12 isotope, as 12 u is the exact atomic mass of that isotope. The atomic mass is usually measured in the units unified atomic mass unit (u), or dalton (Da). These concepts are further explained below. Mass of molecule calculated from the mass of its isotopes (in contrast of measured ba a mass spectrometer) Integer mass of molecule consisting of most abundant isotopes Ratio of mass m of a molecule and and the atomic mass constant m u Ratio of mass m and and the atomic mass constant m u Mass Concepts in Chemistry name in chemistry Note that the former is now often referred to as the "molecular weight" or "atomic weight". The former usually implies a certain isotopic distribution, whereas the latter usually refers to the most common isotope ( 16O 2). For example, the macroscopic mass of oxygen (O 2) does not correspond to the microscopic mass of O 2. This means that from a physical stand point, these mixtures are not pure. On the macroscopic level, most mass measurements of pure substances refer to a mixture of isotopes. In addition, the situation is rendered more complicated by the isotopic distribution. The name "atomic mass" is used for historical reasons, and originates from the fact that chemistry was the first science to investigate the same physical objects on macroscopic and microscopic levels. "Neither the name of the physical quantity, nor the symbol used to denote it, should imply a particular choice of unit." 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
